Cerebral microbleeds (CMBs) are small (<10 mm), rounded, homogeneous, hypointense lesions revealed on T2*-weighted gradient-echo (GRE) or susceptibility weighted imaging (SWI) sequences, suggestive of extravasation of blood components through pathologically fragile small vessels.1 Conventionally, CMBs have been considered asymptomatic. However, there have been a few recent reports supporting that acutely developing CMBs may cause clinical symptoms mimicking lacunar infarction.2-7 Herein, we present a case of pure sensory stroke caused by an acute symptomatic CMB with perilesional edema.
CASE REPORT
A 48-year-old woman developed sudden paresthesia in the left hemibody 15 hours prior to admission. Her sensory symptoms started at the left corner of the mouth and the left first two fingers. The oral symptom gradually spread to the face, while the cheiral symptom progressed up to her shoulder over a 60-minute period. A day after symptom onset, she complained of paresthesia in the left thigh and reduced sensation in the left tongue and gum. The patient had a previous history of hypertension and kidney/pancreas transplantation owing to type 1 diabetes and renal failure. Although her medication included a calcium-channel blocker, her blood pressure was poorly controlled, and the systolic blood pressure occasionally rose to ≥200 mmHg.
Upon neurologic examination, there was no weakness, ataxia, or visual field defect. She complained of paresthesia and hypesthesia with symptoms of greater severity in the regions where the symptoms began while the numbness was milder in the regions where the symptoms developed later. The National Institutes of Health Stroke Scale score was 1.
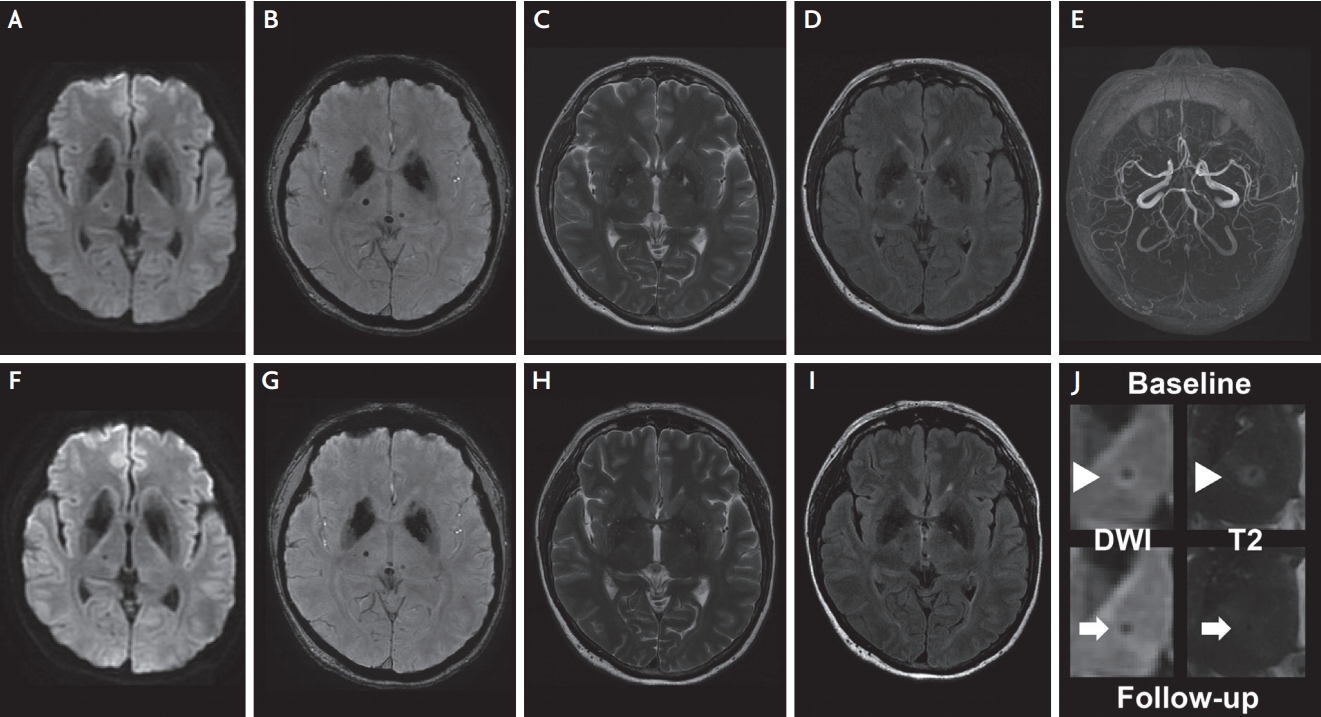
Initial non-contrast brain computed tomography did not reveal any acute lesion. Brain magnetic resonance (MR) image performed 19 hours after symptom onset showed no diffusion restriction lesion on the diffusion-weighted image (Fig. 1A). However, a CMB was noted on the right inferolateral thalamus on the SWI, which was surrounded by a hyperintense rim on the T2-weighted and fluid attenuated inversion recovery (FLAIR) images (Fig. 1B-D), suggestive of acute CMB with perilesional edema.2 Several other CMBs in the left basal ganglia, thalamus and pons were also observed on the SWI (Fig. 1B), but these lesions did not show adjacent parenchymal edema on the T2-weighted image (Fig. 1C). MR angiography showed no significant intracranial arterial stenosis (Fig. 1E).
The patient was diagnosed with an acute symptomatic CMB in the right inferolateral thalamus. Her symptoms resolved over time following the exact reverse order of symptom development. Seven days later, she reported mild hypesthesia only in the first two digits of her left hand, which gradually resolved over months. The patient underwent follow-up brain MR imaging 5 months after the symptom onset (Fig. 1F-I). The size of previous symptomatic CMB decreased slightly on the SWI, and the hyperintense rim disappeared on the follow-up DWI and T2-weighted images (Fig. 1J).
DISCUSSION
In general, CMBs have been considered asymptomatic lesions.1 However, a few recent reports reveal that CMBs can cause clinical symptoms that mimic cerebral ischemia, as in our case.2-7 In a recent study investigating the incidence of suspected symptomatic CMBs in diffusion-negative stroke patients, seven out of 14 (50%) patients whose symptom persisted for more than 24 hours showed CMBs surrounded by hyperintense rims on their MR images, implying that a certain proportion of diffusion-negative stroke may be explained by symptomatic CMBs.6 However, ascertaining whether the symptoms are indeed attributable to the CMBs is challenging as the temporal causal relationship between CMBs and neurological symptoms is difficult to prove.2,6,7 Several reports have claimed that the existence of hyperintense rim surrounding the CMB could be useful in differentiating acute symptomatic CMBs from chronic CMBs.2 Contrary to chronic CMBs, newly-developed symptomatic CMBs tend to be surrounded by a perilesional hyperintense rim on a T2-weighted image, considered vasogenic edema due to microhemorrhage.2-4 Thus, the CMB-induced acute stroke syndrome may possibly be explained by perilesional edema.2,8
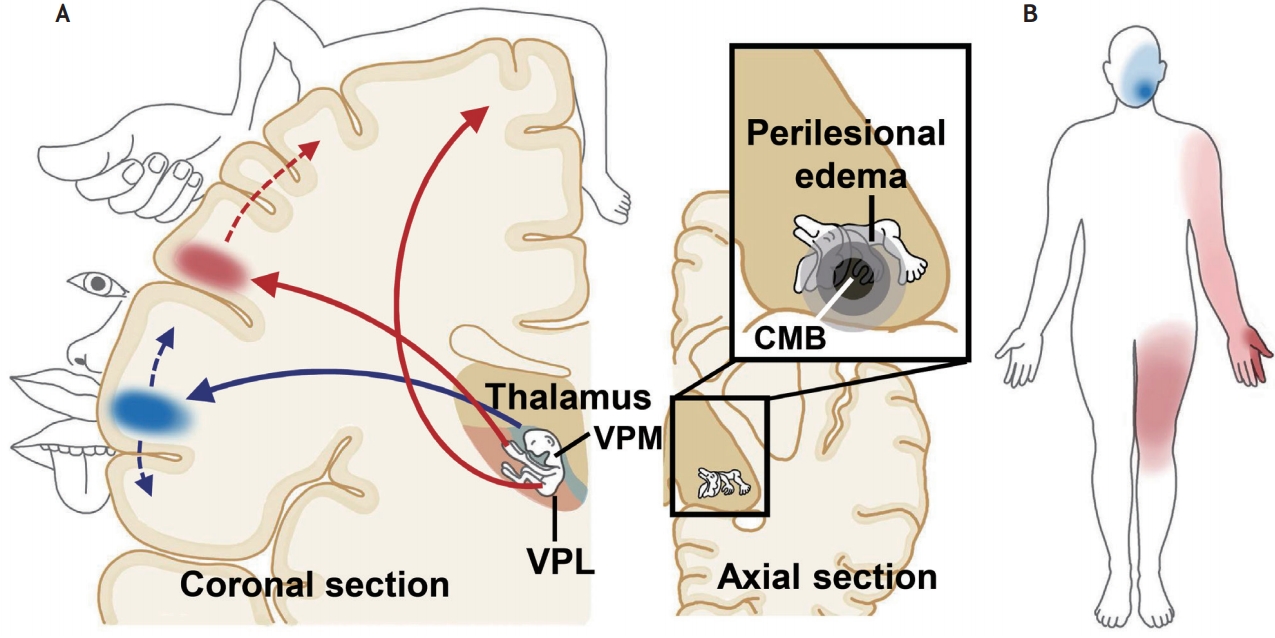
The patient described in this report supports this perspective. It can be inferred that the patient’s CMB developed in the junction of the ventroposterolateral and ventroposteromedial nucleus, causing cheiro-oral symptoms due to close mapping sensory homunculi in the thalamus,9,10 and extension of the sensory symptoms to the left hemibody was attributed to gradual expansion of the perilesional edema to a wider portion of thalamic sensory homunculi (Fig. 2). Intriguingly, the symptoms resolved following the reverse order of initial appearance and extension, which may possibly be attributed to shrinkage of perilesional edema over time. This finding holds clinical implication in that CMBs can even mimic stroke progression.
Given that the patient did not undergo MR imaging prior to the onset of symptoms, we cannot assure that the right thalamic CMB observed was indeed an acute symptomatic CMB as it could also be a chronic pre-existing CMB. However, considering that her initial MR images revealed symptom-correlating right thalamic CMB (SWI) surrounded by hyperintense rim (DWI, T2, and FLAIR) and that the hyperintense rim disappeared on the follow-up MR image, it is more likely that the CMB as a newly-developed CMB rather than a chronic one.
Clinicians should be aware that acute stroke syndrome can also be caused by CMBs, and symptoms can worsen with expansion of perilesional edema. Early MR image acquisition, including the T2*-weighted GRE or SWI sequences, may be informative in planning treatment strategies, especially considering the high risk of hemorrhage in patients with CMBs.