 |
 |
- Search
| J Neurosonol Neuroimag > Volume 10(2); 2018 > Article |
|
Abstract
Background
Cerebral autosomal-dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) is the most common genetic disorder causing stroke. Cerebral microbleeds (CMB) are known to be associated with CADASIL. In this study, we investigated the spatial distribution of CMB in different age groups and also elucidated the age-related spatial distribution of CMB according to the presence or absence of hypertension in patients with CADASIL.
Methods
Ninety-four patients with genetically conf irmed CADASIL and the R544C, R578C, R75P, and C452A mutations were investigated. The patients were divided into three groups according to age: <60 years (n=37), 60-69 years (n=24), and ≥70 years (n=33). The location of the CMB was classified as deep, lobar, or infra-tentorial.
Results
The patients aged <60 years had a significantly higher rate of hypercholesterolemia, a lower rate of hypertension, and a lower rate of use of antiplatelet agents than the patients aged ≥60 years. The group aged ≥70 years had significant more lobar CMB than the group aged <60 years. However, there was no significant difference in the number of patients with lobar CMB between the hypertensive subgroup aged ≥60 years and the hypertensive subgroup aged <60 years. There was no signif icant difference in the numbers of deep or infra-tentorial CMB among the three age groups.
Cerebral autosomal-dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) is the most common inherited cerebral small vessel disease and is caused by mutations in the NOTCH3 gene [1]. Transient ischemic attacks and ischemic strokes are the most frequent manifestations of CADASIL [2]. Moreover, CADASIL predisposes to the development of cerebral microbleeds (CMB) [3,4]. CMB are well defined brain lesions that are demonstrable on magnetic resonance imaging (MRI) and consist of tiny hemosiderin deposits surrounding macrophages adjacent to the abnormal small vessel. They are detectable using T2*-weighted gradient echo or susceptibility-weighted imaging (SWI) [5].
Several studies have reported that CMB might be relevant to the risk of intracerebral hemorrhage (ICH), which results in functional dependence, and may even predict ICH. Hypertension and age are known to be associated with the development of CMB [6,7]. However, the distribution and characteristics of CMB according to age have not been analyzed in patients with CADASIL [8,9]. Therefore, we sought to elucidate the spatial distribution of CMB according to age and the age-related spatial distribution of CMB according to the presence or absence of hypertension.
The study included 102 consecutive patients with CADASIL from 83 families who attended the Department of Neurology, Jeju National University Hospital between March 2012 and February 2015. The study was approved by the Institutional Review Board of Jeju National University Hospital and informed consent was obtained from patients.
Both asymptomatic and symptomatic patients were included. Symptomatic patients had been diagnosed with CADASIL before the study based on their symptoms (ischemia or hemorrhagic episode, cognitive impairment, chronic headache, and seizure). Asymptomatic patients had at least one symptomatic family member. Eight of the 102 patients with CADASIL were excluded because they had been diagnosed by skin biopsy only, leaving 94 genetically confirmed patients from 76 unrelated families available for analysis. The mutation was R544C in 89 patients, R578C in two patients, R75P in two patients, and C452A in one patient. Most patients with CADASIL start to develop symptoms in their 30s, and are unable to live independently because of walking disorders by 60 years of age, and most die in their 70s. Therefore, in order to observe changes in CMB according to clinical features, the patients were divided into three groups according to age: [2] <60 years (n=37), 60-69 years (n=24), and ≥70 years (n=33). The location of the CMB was classified as deep, lobar, or infra-tentorial.
Vascular risk factors were recorded, including hypertension, diabetes mellitus, and hypercholesterolemia. Hypertension was defined as a blood pressure >140/90 mmHg on at least two clinic-based occasions or use of antihypertensive agents. Diabetes mellitus was defined as a fasting glucose level ≥126 mg/dL, a PP2 test level ≥200 mg/dL, or use of anti-diabetes medication. Hypercholesterolemia was defined as a total serum cholesterol level >240 mg/dL.
All patients had an MRI study acquired on a 3-T scanner (Achieva, Philips Healthcare, Best, the Netherlands) using a 32-channel array head coil. The details of the MRI studies performed in these patients have been reported previously [4]. SWI was performed to evaluate microbleeds. One experienced neuroradiologist (H.K.L.), who was blinded to the clinical data, reviewed all the MRI studies and rated the definite presence, number, and distribution of CMB on SWI using the Microbleed Anatomical Rating Scale (MARS) [10]. According to the MARS, definite CMB was defined as small, rounded or circular, well-defined hypodense lesions within the brain parenchyma with clear margins ranging from 2 mm to 10 mm in size on T2*-weighted images [10] or SWI [11], and the locations of the CMB were classified as deep, lobar, or infra-tentorial. CMB mimics were carefully excluded using all available images.
The statistical analyses were performed using SPSS for Windows (version 20; IBM Corp., Armonk, NY, USA). The differences in demographics and modifiable vascular risk factors among the three age groups were assessed using analysis of variance (ANOVA) and the chi-squared test. The numbers of CMB had a rightskewed distribution so were log-transformed to allow for comparison by ANOVA. To provide additional information about the age-related spatial distribution in the three age groups, we also undertook analyses of the patients grouped by the presence (Fig. 1) of absence (Fig. 2) of hypertension. A p-value ≤0.05 was defined as statistically significant.
The demographic data for the overall study population have recently been published [4]. The mean patient age was 62.6 years and 55% were men. Patients aged <60 years had a significantly higher rate of hypercholesterolemia, a lower rate of hypertension, and a lower rate of use of antiplatelet agents than those aged ≥60 years. Previous use of antiplatelet therapy was significantly more common in the patients aged ≥60 years. There was no statistically significant between-group difference in sex distribution or the rates of diabetes mellitus, atrial fibrillation, ever-smoking, or use of statin medication (Table 1).
Patients aged ≥60 years had more total CMB than those aged <60 years but the difference did not reach statistical significance (p=0.055). There was no significant difference in the total number of CMB between the group aged 60-69 years and the group aged ≥70 years. Lobar CMB was significantly more common in patients aged ≥70 years than in younger patients. There was no significant difference in the number of deep or infra-tentorial CMB among the three age groups (Table 2). There was a trend for a difference in the number of lobar CMB among the three normotensive age subgroups (p=0.059), but this did not reach statistical significance. There was also no significant difference in the age-related spatial distribution of CMB among the three hypertensive age subgroups (Table 3).
We found that patients aged ≥60 years with CADASIL were significantly more likely to have lobar CMB than their younger counterparts. We also identified a trend for a difference in the number of lobar CMB among the three normotensive age subgroups (p=0.069) that did not reach statistical significance. Furthermore, there was no significant difference in the number of lobar CMB among the three hypertensive age groups.
Unlike in an earlier study [4] in which we found a significant difference in the number of lobar CMB between patients aged ≥60 years and those aged <60 years but not in the number of deep or infra-tentorial CMB, our more recent results demonstrated no association between age and presence of lobar CMB [3]. This discrepancy may in part reflect the different assessment methods used in the two mentioned studies. First, we have now used more sensitive MRI techniques to detect CMBs than those used previously: the effects of MRI sequences (traditional T2*-weighted gradient echo vs. SWI); section thickness (5 mm vs. 2 mm); and field strength (1.5 tesla vs. 3 tesla). Second, in the present study, we used the MARS [10] system to calculate CMB. However, in our previous study [3], we divided CMBs into five groups according to brain location (cortical-subcortical, basal ganglia, thalamus, brain stem, and cerebellum) and then calculated the number of CMBs. In addition, SWI, smaller section thickness, and a higher magnetic field are known to increase the number of CMBs detected [12].
In the present study, there was no significant difference in the number of lobar CMB among the three hypertensive age subgroups. However, there was a trend for a difference in number of lobar CMB among the three normotensive age subgroups (p=0.059). This is in line with a previous report of an independent association between age and the presence of CMB when only when normotensive patients were analyzed [9].
The risk factors for CMB vary according to their location in the brain. Lobar CMB is mainly associated with smoking, low cholesterol levels, amyloid angiopathy, and degenerative disease, whereas deep CMB is usually associated with hypertension [6,7]. As reported in this study, patients with CADASIL show an increase in lobar CMB with increasing age. Importantly, lobar CMB is known to be closely related to the development of ICH. Moreover, there have been reports of ICH in patients with CADASIL after antiplatelet therapy. Therefore, the efficacy and safety of anti-thrombotic medication in these patients is unclear [13], and care should be taken when prescribing anti-thrombotic agents in older patients with CADASIL.
We also found no significant difference in the number of lobar CMB in the patients with CADASIL aged 60-69 years and those aged ≥70 years. There is a paucity of data on the presence and number of CMB in Caucasian patients with CADASIL who are aged ≥60 years because these patients are not often able to walk by the age of 60 years and are bed-ridden at around 65 years of age [14,15]. Therefore, we cannot exclude the possibility that there is an age-related increase in the number of CMB after the age of 60 years. However, our present results suggest that the impact of age on the number of CMB is significantly less than that of hypertension, especially beyond the age of 60 years. The one previous study that examined the prevalence of CMB according to location and age in patients with CADASIL enrolled only 40 patients and did not include a statistical analysis [15]. To our knowledge, this is the first study to examine the spatial distribution of CMB according to age group in patients with CADASIL using statistical analysis.
Our study has several limitations. First, patients aged <60 years had a higher rate of hypercholesterolemia and were less likely to use antiplatelet agents than those aged ≥60 years, which could have influenced the number of CMB. However, our recent study showed that hypercholesterolemia was not a predictor of CMB in univariate analysis and that antiplatelet therapy was not a predictor of CMB in multivariate analysis [4]. Second, because the number of normotensive patients with CADASIL was relatively low, this study did not have enough statistical power to demonstrate a significant difference in number of lobar CMB among the three normotensive age subgroups. Finally, an R544C mutation in exon 11 accounted for 95% of the mutations. Therefore, our findings may not be generalizable to patients with other types of mutation for CADASIL [16]. However, the genotype-phenotype correlation in CADASIL remains unclear [17].
In conclusion, our results suggest that patient age may have a limited impact on the number of lobar CMB in patients with CADASIL who are hypertensive. Further prospective studies are needed to elucidate the relationship between age and number of CMB.
Acknowledgments
This work is attributed to and was solely supported by the Department of Neurology, Jeju National University Hospital, Jeju, Korea.
The research was supported by the 2018 scientific promotion program funded by Jeju National University.
Fig. 1.
Susceptibility-weighted images for three normotensive patients with CADASIL. Panels (A-C) are images for patient 1 (aged 39 years), panels (D-F) are images for patient 2 (aged 62 years), and panels (G-I) are images for patient 3 (aged 76 years). The arrows indicate cerebral microbleeds. CADASIL; cerebral autosomal-dominant arteriopathy with subcortical infarcts and leukoencephalopathy.
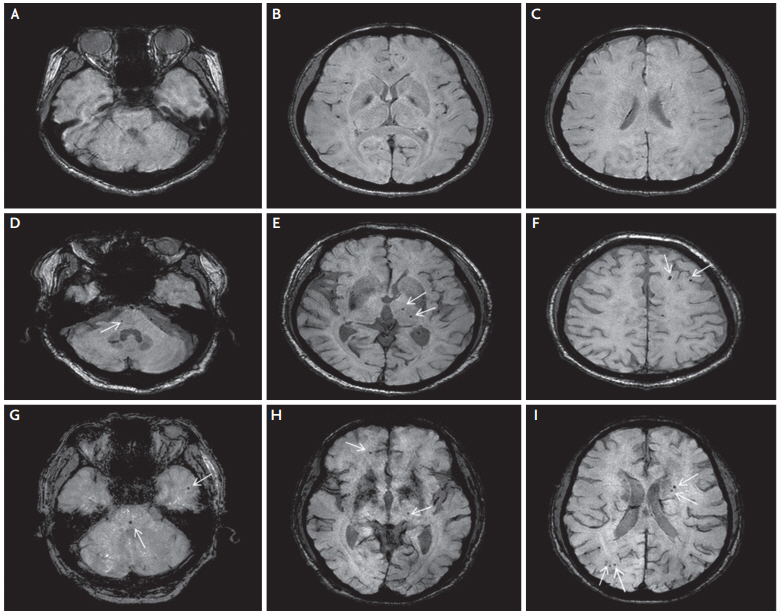
Fig. 2.
Susceptibility-weighted images for three hypertensive patients with CADASIL. Panels (A-C) are images for patient 4 (aged 53 years), panels (D-F) are images for patient 5 (aged 64 years), and panels (G-I) are images for patient 6 (aged 77 years). The arrows indicate cerebral microbleeds. CADASIL; cerebral autosomal-dominant arteriopathy with subcortical infarcts and leukoencephalopathy.
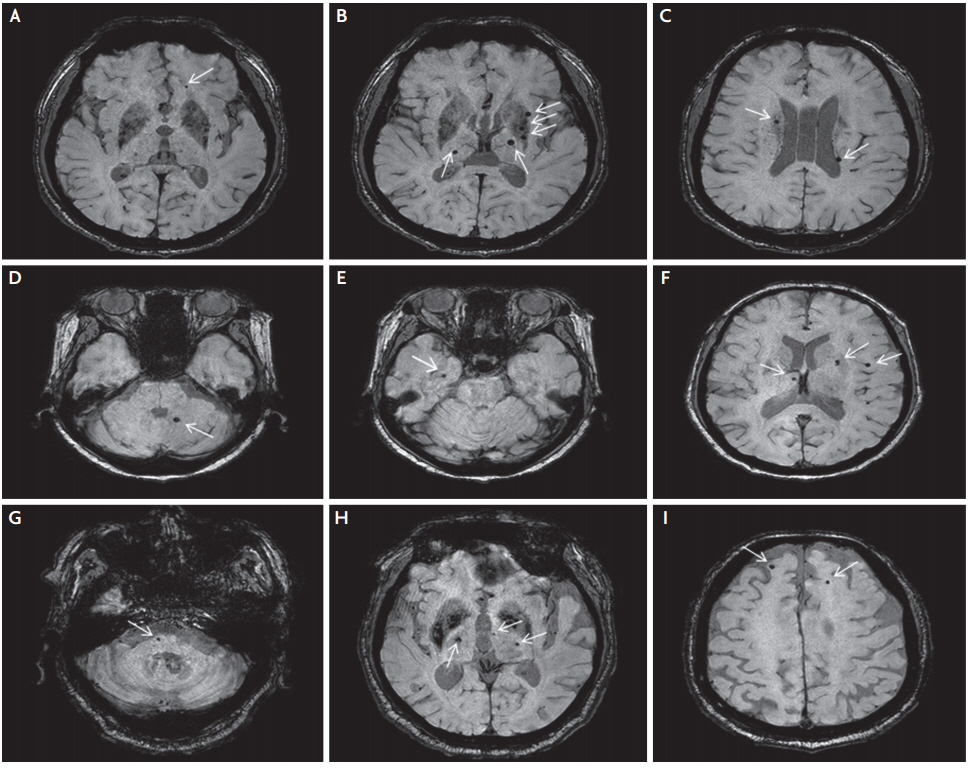
Table 1.
Clinical features of 94 patients with CADASIL and R544C mutation
Table 2.
Numbers of CMB according to age group
Table 3.
Number and location of cerebral microbleeds according to the presence or absence of hypertension
REFERENCES
1. Joutel A, Corpechot C, Ducros A, Vahedi K, Chabriat H, Mouton P, et al. Notch3 mutations in CADASIL, a hereditary adult-onset condition causing stroke and dementia. Nature. 1996;383:707-710.



2. Chabriat H, Joutel A, Dichgans M, Tournier-Lasserve E, Bousser MG. Cadasil. Lancet Neurol. 2009;8:643-653.


3. Lee JS, Kang CH, Park SQ, Choi HA, Sim KB. Clinical significance of cerebral microbleeds locations in CADASIL with R544C NOTCH3 Mutation. PLoS One. 2015;10:e0118163.



4. Lee JS, Ko K, Oh JH, Park JH, Lee HK, Floriolli D, et al. Cerebral microbleeds, hypertension, and intracerebral hemorrhage in cerebral autosomal-dominant arteriopathy with subcortical infarcts and leukoencephalopathy. Front Neurol. 2017;8:203.



5. Greenberg SM, Vernooij MW, Cordonnier C, Viswanathan A, Al-Shahi Salman R, Warach S, et al. Cerebral microbleeds: a field guide to their detection and interpretation. Lancet Neurol. 2009;8:165-174.



6. Kim BJ, Lee SH. Cerebral microbleeds: their associated factors, radiologic findings, and clinical implications. J Stroke. 2013;15:153-163.



7. Kim BJ, Yoon Y, Sohn H, Kang DW, Kim JS, Kwon SU. Difference in the location and risk factors of cerebral microbleeds according to ischemic stroke subtypes. J Stroke. 2016;18:297-303.




8. Lesnik Oberstein SA, van den Boom R, van Buchem MA, van Houwelingen HC, Bakker E, Vollebregt E, et al. Cerebral microbleeds in CADASIL. Neurology. 2001;57:1066-1070.


9. Viswanathan A, Guichard JP, Gschwendtner A, Buffon F, Cumurcuic R, Boutron C, et al. Blood pressure and haemoglobin A1c are associated with microhaemorrhage in CADASIL: a two-centre cohort study. Brain. 2006;129(Pt 9):2375-2383.


10. Gregoire SM, Chaudhary UJ, Brown MM, Yousry TA, Kallis C, Jäger HR, et al. The microbleed anatomical rating scale (MARS): reliability of a tool to map brain microbleeds. Neurology. 2009;73:1759-1766.


11. Shams S, Martola J, Cavallin L, Granberg T, Shams M, Aspelin P, et al. SWI or T2*: which MRI sequence to use in the detection of cerebral microbleeds? The Karolinska Imaging Dementia Study. AJNR Am J Neuroradiol. 2015;36:1089-1095.



12. Nandigam RN, Viswanathan A, Delgado P, Skehan ME, Smith EE, Rosand J, et al. MR imaging detection of cerebral microbleeds: effect of susceptibility-weighted imaging, section thickness, and field strength. AJNR Am J Neuroradiol. 2009;30:338-343.



13. Di Donato I, Bianchi S, De Stefano N, Dichgans M, Dotti MT, Duering M, et al. Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) as a model of small vessel disease: update on clinical, diagnostic, and management aspects. BMC Med. 2017;15:41.




14. Opherk C, Peters N, Herzog J, Luedtke R, Dichgans M. Long-term prognosis and causes of death in CADASIL: a retrospective study in 411 patients. Brain. 2004;127(Pt 11):2533-2539.


15. van den Boom R, Lesnik Oberstein SA, Ferrari MD, Haan J, van Buchem MA. Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy: MR imaging findings at different ages--3rd-6th decades. Radiology. 2003;229:683-690.


-
METRICS

-
- 1 Crossref
- Scopus
- 4,689 View
- 66 Download
- Related articles in AC





